Stay Updated with Wellness Digestive




No spam, no fluff—just practical, evidence-based advice delivered straight to your inbox every Tuesday.






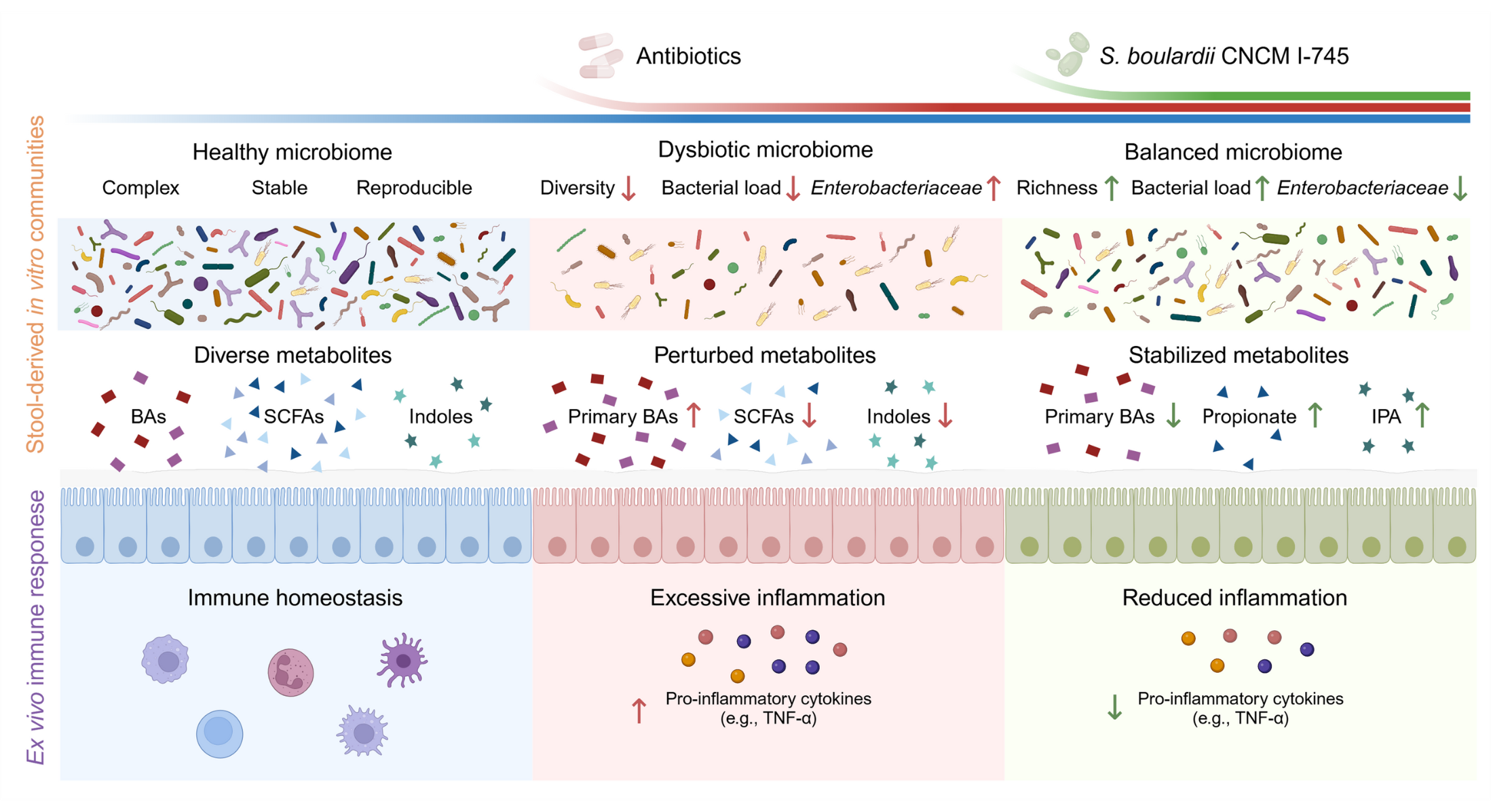
Antibiotic treatments play a vital role in contemporary medical practice, yet they rank among the most significant disruptors of the human intestinal microbial community. These medications do more than just reduce the variety of microbes present; they also break down the complex metabolic networks that are essential for upholding balance within the gut environment. The key challenge in the field of microbiome-based treatments has shifted toward reinstating these critical functions, moving beyond the simple task of repopulating the gut with bacteria.
A groundbreaking investigation conducted by Huang and colleagues, published in 2025 in Gut Microbes, marks a pioneering effort in this area. For the first time, researchers have shown that the probiotic yeast Saccharomyces boulardii CNCM I-745 not only helps maintain the stability of the gut microbiota composition when exposed to antibiotics but also effectively reinstates its metabolic capabilities. This preservation ensures the ongoing biochemical interactions between the microbial community and the host remain intact.
The study employed an innovative combination of sophisticated in vitro models simulating the human gut, along with metagenomic sequencing, metabolomic profiling, and assays involving human immune cells. Through this multifaceted approach, the scientists were able to isolate and examine the yeast’s direct influences on the microbiota, without interference from host-related factors. Such a comprehensive methodology sets a new standard that can be adopted for evaluating other probiotics aimed at restoring functional aspects of the gut ecosystem.
Historically, research on probiotics has emphasized changes in the taxonomic makeup of the microbiota, monitoring shifts in the abundance of particular microbial groups in response to interventions. Nevertheless, the true indicator of a robust gut microbial ecosystem is its functional performance: the capacity of these microbes to break down dietary fibers, generate advantageous metabolites, and help maintain immune equilibrium.
In their work, Huang et al. utilized advanced quantitative profiling of the microbiota alongside shotgun metagenomics to thoroughly document both the structural changes and the functional alterations within the microbiome when subjected to antibiotic challenges. The experiments were carried out using two distinct in vitro human gut microbiota cultivation systems: the static MiPro model and the dynamic SHIME system. Within these setups, the commonly prescribed antibiotic combination amoxicillin/clavulanic acid caused a substantial decline in bacterial numbers, species diversity, and overall metabolic productivity. However, when S. boulardii CNCM I-745 was added as a supplement, it successfully counteracted these disruptions by sustaining bacterial populations and reactivating essential metabolic routes involved in energy production and carbohydrate processing.
This observation holds particular importance because it reveals that simply protecting the existing microbial biomass-without needing to overhaul the community structure-is often enough to uphold the operational wholeness of the gut ecosystem. The probiotic yeast functioned like a supportive anchor, enabling the native microbial residents to persist in their vital roles despite the antibiotic-induced pressure.
One of the standout elements of this research is its in-depth metabolomic examination, which provided granular insights into microbial outputs. Exposure to antibiotics severely impaired the synthesis of short-chain fatty acids (SCFAs) and metabolites derived from tryptophan, both of which are pivotal in facilitating communication between the microbiota and the host organism.
Supplementing with Saccharomyces boulardii CNCM I-745 successfully brought back the levels of propionate and indole-3-propionic acid (IPA), two key metabolites that play complementary roles in gut health. Propionate, primarily generated by Bacteroides species as an SCFA, plays a crucial part in shaping mucosal immune responses and fostering the growth of regulatory T cells. Meanwhile, IPA, which arises from the metabolism of tryptophan, bolsters the strength of the intestinal epithelial barrier and suppresses inflammation driven by the NF-κB pathway.
Notably, the yeast S. boulardii itself exhibited very limited metabolic activity in oxygen-free environments, which points to the idea that its benefits arise through ecological support mechanisms. Rather than producing these metabolites on its own, the yeast enhances the recovery and functionality of the indigenous bacterial populations. This indirect, facilitative strategy could well be a hallmark characteristic that distinguishes probiotic yeasts from other microbial interventions.
To bridge the gap between microbial effects and host responses, the research team tested supernatants derived from the microbiota cultures in the MiPro and SHIME models on human peripheral blood mononuclear cells (PBMCs) and intestinal mucosal tissue samples obtained from healthy individuals. Supernatants from microbiota solely exposed to antibiotics provoked a robust release of pro-inflammatory cytokines, such as TNF-α, IL-6, and MCP-1. On the other hand, supernatants from microbiota supplemented with S. boulardii showed a dramatic reduction in these inflammatory markers.
Intriguingly, direct application of S. boulardii supernatant to human cells had negligible impact on cytokine levels, further emphasizing the indirect nature of its immunomodulatory benefits. These results indicate that S. boulardii influences the immune system primarily by reinvigorating the microbiome’s metabolic profile. Through the sustained production of anti-inflammatory compounds like SCFAs and IPA, the yeast fosters a gut milieu that supports immune tolerance and facilitates the repair of mucosal tissues.
This clear delineation of mechanisms bolsters the case for therapies centered on microbial metabolites as companions to antibiotic regimens. It also deepens our comprehension of the interconnected pathway linking microbiota, their metabolic byproducts, and immune function in promoting gut resilience.
A major strength of this study resides in its methodological sophistication and precision. By seamlessly combining static in vitro systems like MiPro with dynamic ones such as SHIME, and integrating them with human cell-based immune assays, the researchers traced the effects of S. boulardii across multiple biological levels-from microbial metabolic processes to tangible host immune reactions.
Crucially, these in vitro human gut microbiota models utilized microbiota sourced directly from humans and deliberately omitted host variables such as dietary influences, psychological stress, concurrent medications, or host cellular components. This controlled design enabled a precise dissection of microbe-to-microbe interactions, free from external confounders. Consequently, the study lays down a solid, reproducible platform that future investigations into probiotics and microbiome therapies can build upon.
The discoveries from Huang et al. resonate widely across the microbiome research landscape. They highlight that probiotics do not necessarily need to induce sweeping changes in microbial community structure to deliver substantial health benefits. Instead, the capacity to sustain microbial biomass and protect core metabolic functions might be the primary factors driving ecosystem resilience.
From this perspective, S. boulardii CNCM I-745 stands out as a representative of advanced probiotics. It protects microbial operations, counters dysbiosis triggered by antibiotics, and aids in achieving host immune homeostasis-all without engaging in direct competition with native bacteria or the antibiotics administered. As the field of microbiome science progresses toward greater precision and a focus on functionality, this research offers an exemplary blueprint for assessing probiotics. It integrates microbial ecology, metabolic pathways, and immunological responses into a cohesive systems biology framework, paving the way for more targeted therapeutic developments.